Mechanics of batteries
- Sumit Basu
- Mar 9
- 10 min read
Updated: Mar 9
The workings of a rechargable batttery --- the lifeline of portable, wireless and sustainable electronics --- depends as much on electro-chemistry as it does on mechanics. The science of lithiation and delithiation makes a durable energy storage device possible but extending its cycle-life requires an understanding of the mechanical connotations of this science. We got five experts, Profs Jeevanjyoti Chakraborty (IIT Kharagpur), Dipayan Mukherjee (IIT Kanpur), Shrinidhi S Pandurangi (IIT Bombay), Siladitya Pal (IIT Roorkee) and Dr Prashant Srivastava (Oorja Bengaluru), to talk about how mechanics can help design better batteries.
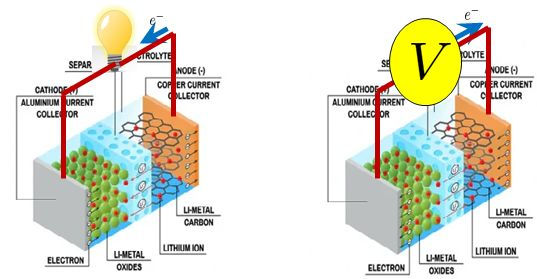
Lithium ion batteries consist of a graphitic anode, a lithium metal oxide (e.g. lithium cobalt oxide, lithium iron phosphate), an electrolyte of a salt of lithium (e.g. lithium hexafluorophosphate) in an organic solvent (e.g. an equal mixture of ethylene carbonate and dimethyl carbonate). The electrolyte floods the two electrodes, which are separated by a microporous polymeric separator that prevents short-circuiting while allowing only lithium ions to pass.
The lithium ion battery is rechargeable and operates in two modes. When a load is connected across its terminals (as in the figure on the left), the battery is operating in the discharging mode. Lithium atoms embedded in the graphite anode oxide into Li+ which travels towards the cathode through the electrolyte and the separator, while electrons produced travel through the external circuit. At the cathode, lithium ions are reduced to lithium atoms by electrons that reach the cathode through the external circuit.
On the other hand, during charging (figure on the right), exactly the reverse happens. Lithium undergoes oxidation at the cathode, drifts towards the anode and gets reduced there by combining with electrons travelling through the external circuit from the cathode to the anode.
Two important points need to be noted here. Firstly, if we adhere to the electrochemical definition of an anode as the electrode where oxidation takes place, in a lithium-ion battery the anode and cathode switch roles during charging and discharging. The graphite electrode acts as the 'anode' during discharge and the lithium metal oxide electrode during charging. However, in the battery community, it is customary to refer to the graphite electrode as the anode and the lithium metal oxide electrode as the cathode in both charging and discharging modes. We will stick to this convention in this article.
Secondly, for the idea of a rechargeable battery to work, lithium must be able to intercalate into both the anode and cathode easily. The chemistry of intercalation is undoubtedly the most remarkable aspect of rechargeable batteries.
A short history
Lithium is the metal of choice for rechargeable batteries because it has one of the most negative potentials with respect to the standard hydrogen electrode, indicating that it is highly prone to oxidation (i.e. losing an electron to become Li+). On top of that, lithium has a small ionic radius that allows it to intercalate into a solid and diffuse in a liquid.
The first attempt to produce a rechargeable battery with a high energy density, based on the idea that certain layered materials allow reversible insertion of molecules in the space between the atomic layers (a phenomenon called intercalation) was initiated at the height of the global oil crisis triggered by the Arab Israel war and the Iranian Revolution of the 1970s.
A British-American chemist, Stanley Whittingham, working for Exxon, used a titanium disulphide cathode and a lithium-aluminium anode to produce the first intercalation-based, reversible battery. In the 1980s, John B. Goodenough at the University of Oxford found that using a lithium cobalt oxide cathode material could almost double the capacity of a Li-ion battery. Finally, in 1985, Akira Yoshino, then working for the Asahi Kasei Corporation in Japan, patented the first commercially viable lithium-ion battery using a carbonaceous cathode. This battery was commercialised by Sony in 1991 and AT&T the following year. Whittingham, Goodenough and Yoshino received the Nobel Prize in 2019 for their discoveries.
The equations behind the working of a Li-ion battery
To understand how mechanics plays a role in the operation of a lithium-ion battery, we first need to understand how lithium ions move back and forth across the cell. The electro-chemo-mechanical processes that unfold during battery charging can be simplified by neglecting the mechanical stresses. Further, because battery processes have timescales of seconds and charge relaxation occurs at much shorter timescales, we can assume continuity of the current density, implying that the current entering any region is equal to that leaving it.
The primary equation of electrostatics that applies is Gauss' law, which states that the electrostatic potential obeys Poisson's equation. Ohmic conduction applies everywhere except in the electrolyte, so that the current density is proportional to the electric field. The electric field, in turn, is given by the negative gradient of the electric potential.
Let us start with the copper collector attached to the anode and move inward toward the cathode. At the copper collector, electrons are the only mobile charged species and conductivity is very high. This means that the current collectors (both the copper and the aluminium ones) are, to a good approximation, equipotential.
Next comes the graphite electrode. In reality, this is not a monolithic block of solid. It is generally a microporous assembly of graphite particles held together by a binder, with the pores flooded with electrolyte.
The graphite particles contain free electrons that, like all current densities, obey the current continuity condition mentioned above. But there is a small twist here to which we will come in a moment.
The graphite particles also have lithium atoms that are unaffected by the electric field but can diffuse into the solid graphite, obeying a possibly non-linear diffusion equation in which the chemical potential gradient controls the flux.
In the electrolyte flooding the pores, recall that the lithium salt has split into Li+ ions and an anion. To determine the concentration of lithium ions, we need to remember that they now drift both against the concentration gradient (i.e., from regions of high to low concentration) and be driven in the direction of the electric field. Also, the current density is due to both Li+ and the anions. In fact, it is important to know how much of the total current is carried by the lithium ions. In real electrolyte solutions where the salt concentration is high and the ion-ion interactions are strong, the 'transference number' must be determined experimentally. The current in the electrolyte is entirely ionic and also obeys the continuity equation.
Some basic interface conditions tie these domains together. The electrostatic potential and the current density are continuous across the interfaces between the current collector and the solid electrode. The situation is a bit more complicated at the interface between the solid electrode and electrolyte. An atomically thin layer (called the Stern layer) of a very high electric field exists at this interface, where electrons and ions line up to form a very thin capacitor. The charge stored in this capacitor helps to maintain a potential difference between the solid electrode and the electrolyte. In many models, this layer is treated as a charged surface across which a jump in the normal component of the electric displacement occurs.
Oxidation or reduction reactions occur on the reaction plane at the outer boundary of the Stern layer. These reactions produce electronic currents in the solid and ionic currents in the electrolyte. The two currents together satisfy continuity. At the reaction plane therefore, they are equal in magnitude and opposite in sign. Thus, when we apply current continuity at the solid electrode, the twist we mentioned earlier involves a non-zero contribution on the right-hand side of the continuity statement, which states that the net current density emanating from a point equals the current transferred into the solid and liquid phases that is now distributed throughout the volume of the electrode particle. Since oxidation produces one lithium ion for each electron, the same argument applies to the continuity equation in the electrolyte except that the right hand side has the opposite sign.
What is the magnitude of the current density produced on the reaction plane? The rates of the oxidation or reduction reactions determine this. The rate, in turn, is influenced by the 'overpotential', which is the deviation of the potential jump across the Stern layer over its equilibrium value. The overpotential drives the Faradaic oxidation or reduction process near the interface.
The electronic current density generated at the reaction plane is determined by the Butler-Volmer equation, which accounts for the forward and reverse reaction rates. This input determines the amount of electronic current injected into the solid and the ionic current injected into the electrolyte.
And where is the mechanics?
Where does the mechanics come into the picture? While charging, lithium ions are reduced at the anode and the resulting lithium atoms intercalate into the carbon anode, driven by the chemical potential gradient [1,2]. The anode swells as a result. If it is mechanically constrained, swelling can generate stresses large enough to cause fracture in the anode. Graphite, for example, swells by about 9-10% (mainly due to lithium atoms forcing an increase in interlayer spacing) as it goes from C6 to LiC6 [3].
The problem is more severe for some candidate non-graphite anodes. For instance, Silicon anodes are under active consideration as a replacement for graphite. These electrodes have approximately ten times the theoretical specific capacity of graphite and are extremely attractive from a technological point of view (the energy density is calculated as 𝑛𝐹/3600𝑀 mAh/g considering that 1 mAh ≃ 3.6 C and 𝐹 = 96485 C/mol. The molar mass of the host is 𝑀, while 𝑛 gives the number of electrons transferred per reaction. For graphite C6+Li++e-→LiC6 makes 𝑛 = 1. The number is 3.75 for Si). However, the major stumbling block is the large volume expansion (~300%) caused by Li alloying with Si. The stresses can be high enough to cause a fracture or plastic deformation. Repeated lithiation and delithiation can cause plastic deformation of Si to occur at every cycle.
Also, stresses and cracks significantly affect charge-discharge responses. Experiments show that the open-circuit potential of the cell is directly affected by applied stress. This points to a connection between stress and the electrostatic potential. The solid-phase diffusion phenomenon can also be influenced by host stress. The chemical potential gradient driving lithium atom diffusion in the host should also depend on stress.
While discharging, the reduction reaction shifts to the cathode. Lithium ions intercalate into the lithium metal oxide electrode. However, this causes much less stress to develop in the oxide, because the size mismatch between the interstices and the intercalating Li atom is not so large. This is expected as the lithium atoms primarily fill the spaces they vacated during the previous charging cycle.
An involved electro-chemo-mechanical problem unfolds inside a battery, and various aspects of this story remain unclear. What is surely known is that the battery's life is limited by the stresses that develop and the fracturing that results.
Open problems
The very large swelling stresses in Si can cause the anode to delaminate from the current collector. Here, the location of the failure plane is pre-determined. But to predict, say, how many cycles it will take for the delamination to make the battery unusable, we need to model the gradual delamination process. A suitable model for such scenarios is the cohesive zone model, which mimics the actual microscopic delamination process by using a traction-separation law that models the degradation of cohesion with increasing separation at the interface. While we know how to incorporate such a cohesive zone into a large-scale Finite Element model of the battery, the challenge is designing and validating the traction-separation law.
Polycrystalline cathodes are commonly used in modern batteries. In these cathodes, very small single-crystalline particles are sintered into larger polycrystalline micrometre-scale particles, which are then held together by a polymeric binder. But the micrometre-scale particles are polycrystalline, with grain boundaries that form during the high-temperature processing route. Metal oxides of lithium like nickel-manganese-cobalt oxide (NMC) or nickel-cobalt-aluminium oxides (NCA) are common polycrystalline cathode. Though intercalation stresses are somewhat lower on the cathode side, the presence of small grains and grain boundaries create highly stressed regions. The grain boundaries are prone to cracking. Again, sophisticated mechanical models that describe the development of these stresses across an ensemble of single-crystal grains are necessary. Even harder is predicting fracture paths that may result from these stresses. Computational techniques, such as the phase field model (PFM), which describe a fracture path as the diffusion of a damage field percolating through the material and do not assume a pre-determined fracture path, are under active consideration.
Can we get rid of the grain boundaries? An idea that leaps to mind is that of a single crystal cathode. While these are hard to manufacture, their mechanics under lithation is still interesting. In fact, mechanics can help determine the optimal single-crystal particle size that is immune to sudden collapse during lithiation/delithation. A candidate material is LNO (lithium nickel oxide) which has a typical layered crystal structure. Single crystals of micrometer dimensions can be grown. But when lithium is removed from the structure during delithiation (during charging), the crystal tends to contract in the direction perpendicular to the layers.
Another important aspect is the nanometer-thick solid electrolyte interface (SEI), which forms at the graphite or silicon anode when the battery is first charged. This layer consists of products of electrolyte decomposition, formed by reduction of electrolyte ions by electrons at the bare anode surface. The layer acts as a thin membrane that allows lithium ions to pass but blocks electrons. Thus, once formed, it stabilises the battery and prevents continued electrolyte decomposition. But remember that the electrolyte actually floods the gaps between the electrode particles, thereby providing a very large surface area. Large deformation of the electrode particles due to intercalation (especially in Si anodes) repeatedly breaks the SEI and reforms it in every cycle, leading to capacity loss. The process is similar to that of metal corrosion. Again, experimentally informed mechanical models for the breaking and reforming of the SEI is an important work in progress.
The above discussion highlights an overarching point. Very large deformation of anode particles is unavoidable in a modern battery, and the fundamental question that all models of the detailed electro-chemo-mechanical processes have to grapple with is the following: how exactly does the system's free energy change with mechanical stress? An experimentally informed answer to this question determines the model's realism.
In fact, the mechanics of lithiation and delithiation do not seem to have yet caught the attention of the battery industry. The primary reason is the difficulty of determining the many hidden parameters in these models. The industry relies on much coarser-grained models that use cycling experiments on entire battery-packs for validation. Battery life is estimated using models built from these experiments.
For particle-level detailed mechanics to be practically useful, they need to address macroscopic issues at the level of an entire battery pack. For instance, the mechanics should translate into predictions of actual puffing of individual units in a battery pack. The puffing of batteries, as the industry well knows, is not directly related to the swelling of electrode particles during lithiation/delithiation, except possibly in the case of Si anodes, where swelling is very large. For most part, puffing results from gas generation due to electrolyte degradation and SEI formation. We have some ground to cover before mechanics can provide definitive answers to practical problems that the battery industry faces.
[1] A. Bower, P. R. Guduru, V. A. Sethuraman, "A finite strain model of stress, diffusion, plastic flow, and electrochemical reactions in a lithium-ion half-cell", J. Mech. Phys. Solids 59, 804-828 (2011)
[2] Z. Cui, F. Gao, J. Qu, "A finite deformation stress-dependent chemical potential and its
applications to lithium ion batteries", J. Mech. Phys. Solids 60, 1280-1295 (2012)
[3] F. Brosa Planella et al. "A continuum of physics-based lithium-ion battery models
reviewed", Prog. Energy 4, 042003 (2022)



Comments